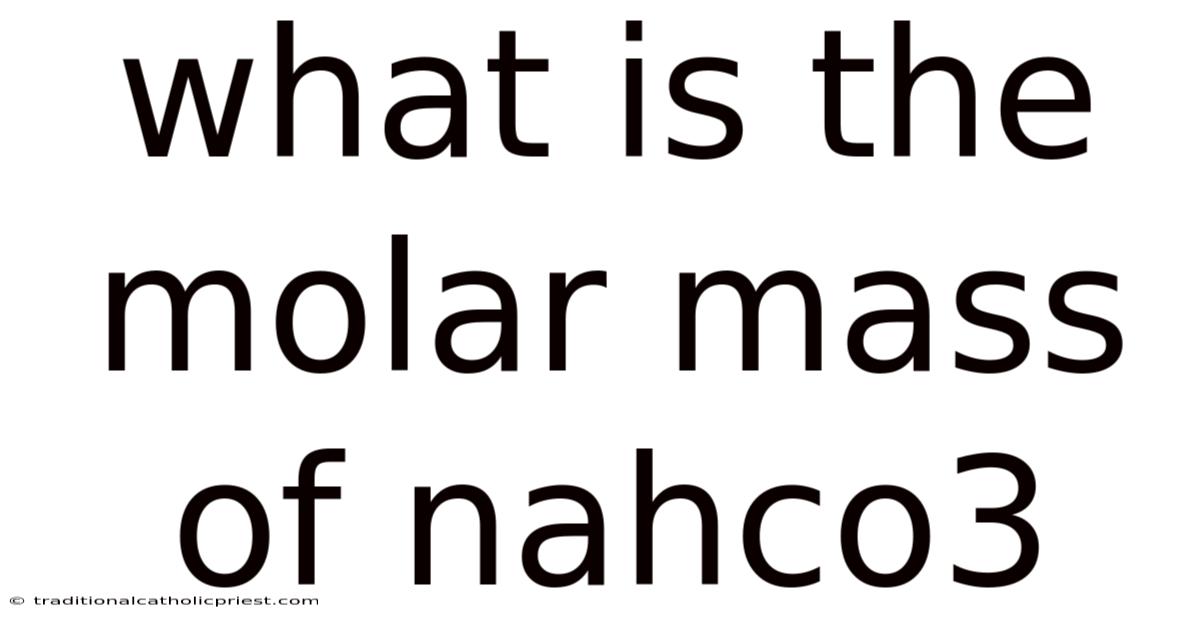Imagine you're in a lab, ready to conduct an experiment, and the success of your entire procedure hinges on precisely measuring out a specific chemical compound. But how much NaHCO3 do you actually need to add to your solution? That said, you glance at the label: NaHCO3, also known as sodium bicarbonate, a common ingredient in baking powder and antacids. This is where understanding the molar mass of NaHCO3 becomes crucial. It's not just about knowing the formula; it's about understanding the fundamental concept that links the microscopic world of atoms and molecules to the macroscopic world we can measure and observe Easy to understand, harder to ignore. Practical, not theoretical..
Calculating the molar mass of any compound is like deciphering a secret code that allows us to convert between mass and moles—a cornerstone in quantitative chemistry. Consider this: whether you're a student, a seasoned researcher, or simply a curious individual eager to grasp the basics of chemistry, understanding how to calculate the molar mass of NaHCO3 is an invaluable skill. This skill empowers you to perform accurate calculations, ensure precision in experiments, and truly comprehend the quantitative relationships that govern chemical reactions. So, let's dive into the world of molar mass and uncover the step-by-step process of determining the molar mass of NaHCO3.
Main Subheading: Grasping the Essence of Molar Mass
At its core, molar mass serves as the bridge between the mass of a substance and the amount of substance, measured in moles. To truly appreciate its significance, we must first understand its definition and importance. Molar mass is defined as the mass of one mole of a substance, whether it be an element, molecule, or compound. It is expressed in grams per mole (g/mol), providing a convenient way to convert between mass and moles. This conversion is particularly useful in stoichiometry, where we need to calculate the amounts of reactants and products in a chemical reaction. Without a clear understanding of molar mass, performing accurate stoichiometric calculations becomes virtually impossible Simple as that..
Imagine trying to bake a cake without measuring the ingredients accurately. The result would likely be a disaster, with the cake either too sweet, too dry, or simply not rising properly. Similarly, in chemistry, using the correct molar masses ensures that you have the right proportions of reactants, leading to successful and predictable outcomes. Still, whether you are synthesizing new compounds, analyzing environmental samples, or formulating pharmaceuticals, the concept of molar mass is indispensable. It provides the foundation for precise measurements and accurate predictions, ensuring that your experiments and calculations are grounded in solid quantitative principles Most people skip this — try not to..
Counterintuitive, but true.
Comprehensive Overview: Diving Deeper into Molar Mass
Definitions and Foundations
Molar mass is the mass of one mole of a substance, typically expressed in grams per mole (g/mol). A mole, in turn, is defined as the amount of a substance that contains as many elementary entities (atoms, molecules, ions, etc.) as there are atoms in 12 grams of carbon-12. This number, known as Avogadro's number (6.Practically speaking, 022 x 10^23), provides a universal standard for counting particles at the atomic and molecular level. The concept of molar mass is rooted in the atomic theory, which states that all matter is composed of atoms, and each element has a unique atomic mass Worth keeping that in mind..
No fluff here — just what actually works.
Scientific Basis
The scientific basis of molar mass lies in the periodic table, where each element is assigned an atomic mass. Atomic mass is the average mass of an atom of an element, expressed in atomic mass units (amu). Also, these values are determined experimentally using mass spectrometry and are based on the relative abundance of isotopes. Practically speaking, the molar mass of a compound is simply the sum of the atomic masses of all the atoms in the compound's chemical formula. This additive property of molar mass allows us to calculate the molar mass of complex molecules and compounds by breaking them down into their constituent elements Practical, not theoretical..
Historical Context
The concept of molar mass evolved alongside the development of modern chemistry. Even so, dalton's atomic theory laid the foundation for understanding that elements combine in fixed ratios to form compounds. In the early 19th century, scientists like John Dalton and Amedeo Avogadro made significant contributions to understanding the relationships between atoms, molecules, and their masses. Avogadro's hypothesis proposed that equal volumes of gases at the same temperature and pressure contain equal numbers of molecules. These ideas, combined with experimental techniques for measuring atomic and molecular masses, led to the development of the concept of molar mass as we know it today Easy to understand, harder to ignore..
And yeah — that's actually more nuanced than it sounds.
Essential Concepts
Understanding molar mass also requires familiarity with related concepts like atomic mass, molecular mass, and formula mass. Because of that, atomic mass refers to the mass of a single atom, typically expressed in atomic mass units (amu). Consider this: molecular mass is the mass of a single molecule, also expressed in amu, and is calculated by summing the atomic masses of all the atoms in the molecule. Plus, formula mass is similar to molecular mass but is used for ionic compounds, which do not exist as discrete molecules. Instead, ionic compounds form a crystal lattice structure, and the formula mass represents the mass of the smallest repeating unit in the lattice And it works..
The Role of Isotopes
Isotopes are variants of an element that have the same number of protons but different numbers of neutrons. Because isotopes have different masses, the atomic mass of an element is actually a weighted average of the masses of its naturally occurring isotopes. Carbon-12 makes up about 98.9% of all carbon atoms, while carbon-13 makes up the remaining 1.Consider this: for example, carbon has two stable isotopes: carbon-12 and carbon-13. In practice, 1%. This weighted average takes into account the relative abundance of each isotope. The atomic mass of carbon is therefore slightly higher than 12 amu, reflecting the small contribution of carbon-13 Small thing, real impact..
Calculating the Molar Mass of NaHCO3
Now, let's get to the practical part: calculating the molar mass of sodium bicarbonate (NaHCO3). This compound consists of one sodium atom (Na), one hydrogen atom (H), one carbon atom (C), and three oxygen atoms (O). To calculate the molar mass, we need to find the atomic masses of each of these elements from the periodic table No workaround needed..
- Sodium (Na): The atomic mass of sodium is approximately 22.99 g/mol.
- Hydrogen (H): The atomic mass of hydrogen is approximately 1.01 g/mol.
- Carbon (C): The atomic mass of carbon is approximately 12.01 g/mol.
- Oxygen (O): The atomic mass of oxygen is approximately 16.00 g/mol.
Now, we multiply the atomic mass of each element by the number of atoms of that element in the chemical formula and then sum the results:
- Molar mass of NaHCO3 = (1 x Atomic mass of Na) + (1 x Atomic mass of H) + (1 x Atomic mass of C) + (3 x Atomic mass of O)
- Molar mass of NaHCO3 = (1 x 22.99 g/mol) + (1 x 1.01 g/mol) + (1 x 12.01 g/mol) + (3 x 16.00 g/mol)
- Molar mass of NaHCO3 = 22.99 g/mol + 1.01 g/mol + 12.01 g/mol + 48.00 g/mol
- Molar mass of NaHCO3 = 84.01 g/mol
So, the molar mass of sodium bicarbonate (NaHCO3) is approximately 84.Now, 01 g/mol. Also, this means that one mole of NaHCO3 weighs 84. Still, 01 grams. This value is crucial for converting between mass and moles in stoichiometric calculations and experimental procedures.
Trends and Latest Developments
The calculation of molar mass remains a fundamental and well-established technique in chemistry. Because of that, one significant trend is the increasing availability and precision of atomic mass data. Modern mass spectrometry techniques allow for highly accurate determination of isotopic abundances, leading to more precise atomic mass values. Still, there are ongoing trends and developments that continue to enhance its accuracy and application. These refined values are regularly updated by international organizations, such as the International Union of Pure and Applied Chemistry (IUPAC), and are incorporated into updated versions of the periodic table.
Some disagree here. Fair enough.
Another trend is the development of computational tools and databases that automate the calculation of molar masses. On the flip side, such tools are particularly useful in fields like drug discovery and materials science, where researchers often work with large numbers of compounds. That's why these tools allow chemists to quickly and accurately determine the molar masses of complex molecules and compounds, without having to manually look up atomic masses and perform calculations. Adding to this, advanced software can account for isotopic distributions, providing even more accurate molar mass values for specific samples.
In recent years, there has also been growing interest in the use of molar mass in fields beyond traditional chemistry. Here's the thing — for example, molar mass plays a critical role in polymer science, where it is used to characterize the size and distribution of polymer chains. Techniques like gel permeation chromatography (GPC) are used to determine the molar mass distribution of polymers, which is essential for understanding their physical and mechanical properties. In nanotechnology, molar mass is used to characterize nanoparticles and other nanoscale materials, providing insights into their composition and behavior.
Tips and Expert Advice
Calculating the molar mass of compounds like NaHCO3 is a fundamental skill, but it's easy to make mistakes if you're not careful. Here are some tips and expert advice to help you master this technique:
1. Double-Check the Chemical Formula: Before you start calculating, make sure you have the correct chemical formula for the compound. A small error in the formula can lead to a significant error in the molar mass. Here's one way to look at it: if you accidentally write Na2CO3 instead of NaHCO3, you will end up with a completely different result. Pay close attention to subscripts and parentheses, and always verify the formula with a reliable source.
2. Use Accurate Atomic Masses: Always use the most accurate atomic masses available, typically from the periodic table provided by IUPAC. While older textbooks may have slightly different values, using the most up-to-date data will ensure the highest level of accuracy in your calculations. Be aware that atomic masses are often given with several decimal places, so make sure to use enough significant figures to match the precision of your experiment or calculation Worth keeping that in mind. No workaround needed..
3. Pay Attention to Units: Remember that molar mass is expressed in grams per mole (g/mol). Make sure to include the units in your calculations and final answer. Including units not only helps you keep track of your work but also ensures that your answer is meaningful and can be properly interpreted Not complicated — just consistent..
4. Practice Regularly: Like any skill, calculating molar mass becomes easier and more intuitive with practice. Work through a variety of examples, starting with simple compounds and gradually progressing to more complex ones. The more you practice, the more confident you will become in your ability to perform accurate calculations.
5. Use Online Calculators as a Check: There are many online molar mass calculators available that can help you check your work. While make sure to be able to perform the calculations manually, using a calculator as a check can help you catch any errors and reinforce your understanding of the process. Just be sure to use a reliable calculator that uses accurate atomic mass data.
FAQ
Q: What is the molar mass of NaHCO3?
A: The molar mass of NaHCO3 (sodium bicarbonate) is approximately 84.01 g/mol.
Q: Why is knowing the molar mass important?
A: Knowing the molar mass is crucial for converting between mass and moles, which is essential in stoichiometry and quantitative chemistry. It allows you to accurately measure out reactants and predict the amounts of products in chemical reactions Simple, but easy to overlook. Surprisingly effective..
Q: Where can I find the atomic masses of elements?
A: You can find the atomic masses of elements on the periodic table. Be sure to use a reliable and up-to-date version of the periodic table, such as the one provided by IUPAC Worth knowing..
Q: How is molar mass different from atomic mass?
A: Atomic mass refers to the mass of a single atom and is expressed in atomic mass units (amu), while molar mass refers to the mass of one mole of a substance and is expressed in grams per mole (g/mol) And that's really what it comes down to..
Q: Can the molar mass of a compound change?
A: The molar mass of a compound is a fixed value based on the atomic masses of its constituent elements. On the flip side, the actual mass of a sample of the compound can vary depending on its purity and the presence of isotopes.
Some disagree here. Fair enough.
Conclusion
Understanding and calculating the molar mass of NaHCO3, or any chemical compound, is a foundational skill in chemistry. Consider this: this concept bridges the gap between the microscopic world of atoms and molecules and the macroscopic world of grams and kilograms that we can measure in the lab. Because of that, by breaking down the chemical formula, identifying the atomic masses of each element, and summing them up, we can accurately determine that the molar mass of NaHCO3 is approximately 84. 01 g/mol.
This changes depending on context. Keep that in mind.
Whether you are a student, researcher, or simply someone with a curiosity for the sciences, mastering this concept will empower you to make accurate measurements, perform reliable calculations, and gain a deeper understanding of the quantitative relationships that govern the world around us. So, take the next step: practice calculating the molar masses of other compounds and explore the fascinating world of stoichiometry. Your newfound knowledge will not only boost your confidence but also open doors to more advanced concepts in chemistry and related fields. Dive into your textbook, try online quizzes, or even set up your own mini-experiment. Share your insights, ask questions, and encourage others to embark on this exciting journey of discovery.