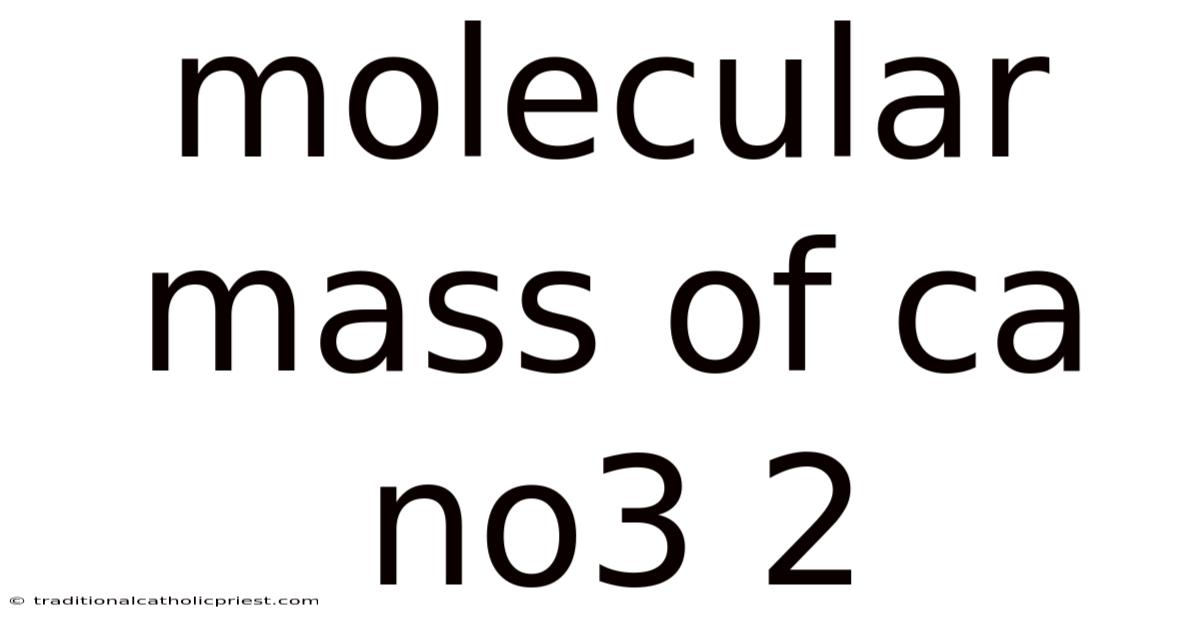Imagine you're in a chemistry lab, carefully measuring out a precise amount of calcium nitrate for an experiment. Understanding the molecular mass of Ca(NO3)2 isn't just a theoretical exercise; it's the foundation for accurate calculations and successful results. Every experiment, every formulation, and every analysis relies on knowing the fundamental properties of the compounds we work with. This knowledge ensures that reactions proceed as expected and that the outcomes are both predictable and reliable.
Think about fertilizers used in agriculture. Calcium nitrate is a key ingredient, providing essential nutrients for plant growth. But how do manufacturers determine the right amount to include in their products? The answer lies in understanding its molecular mass. Without this knowledge, the application rates would be wildly inaccurate, leading to either nutrient deficiencies or environmental harm. The molecular mass serves as the bridge between the microscopic world of atoms and the macroscopic world of measurable quantities, impacting fields from agriculture to medicine.
Demystifying the Molecular Mass of Calcium Nitrate [Ca(NO3)2]
The molecular mass, also known as the molecular weight, is the sum of the atomic masses of all the atoms in a molecule. On top of that, for calcium nitrate, represented by the chemical formula Ca(NO3)2, this means adding up the atomic masses of one calcium atom (Ca), two nitrogen atoms (N), and six oxygen atoms (O). Grasping this concept is fundamental not only for chemistry students but also for professionals who work with chemical compounds daily Not complicated — just consistent..
To calculate the molecular mass accurately, we rely on the periodic table. The periodic table provides the atomic masses of individual elements, which are typically expressed in atomic mass units (amu) or Daltons (Da). By using these values, we can systematically determine the molecular mass of any compound, ensuring that our calculations are precise and our understanding is complete.
Comprehensive Overview: The Essence of Molecular Mass
Defining Molecular Mass
Molecular mass is the mass of a given molecule, measured in atomic mass units (amu). It is numerically equal to the molar mass, but expressed in different units. The molar mass is the mass of one mole of a substance, typically expressed in grams per mole (g/mol). Both terms are often used interchangeably in chemistry, but understanding the distinction is crucial for accurate scientific communication.
The concept of molecular mass is intrinsically linked to Avogadro's number (approximately 6.On top of that, 022 x 10^23), which represents the number of atoms, molecules, or ions in one mole of a substance. Knowing the molecular mass allows us to convert between mass and moles, enabling us to perform stoichiometric calculations and understand chemical reactions at a quantitative level And that's really what it comes down to..
The official docs gloss over this. That's a mistake The details matter here..
Scientific Foundations
The calculation of molecular mass is rooted in Dalton's atomic theory, which posits that all matter is composed of atoms and that atoms of a given element are identical in mass. While isotopes introduce slight variations in atomic mass, the values listed on the periodic table represent the average atomic mass, weighted by the natural abundance of each isotope Still holds up..
Mass spectrometry is an experimental technique used to determine the molecular mass of compounds with high precision. But in mass spectrometry, molecules are ionized and then separated based on their mass-to-charge ratio. Here's the thing — the resulting spectrum provides valuable information about the molecular composition and structure of the compound. This technique is invaluable in fields such as proteomics, metabolomics, and drug discovery The details matter here..
History and Evolution of the Concept
The concept of atomic and molecular masses evolved gradually over centuries. John Dalton's work in the early 19th century laid the foundation for modern atomic theory. Later, scientists like Stanislao Cannizzaro refined these ideas and established a consistent method for determining atomic weights Not complicated — just consistent..
The development of the periodic table by Dmitri Mendeleev in 1869 provided a systematic way to organize elements based on their properties, including atomic mass. The periodic table has become an indispensable tool for chemists, providing a wealth of information about the elements and their compounds. The standardization of atomic masses and the definition of the mole as a unit of measurement further solidified the concept of molecular mass in chemical calculations.
Essential Concepts: Calcium Nitrate [Ca(NO3)2] Breakdown
Calcium nitrate [Ca(NO3)2] is an inorganic compound with a wide range of applications. It consists of one calcium ion (Ca2+) and two nitrate ions (NO3-). To calculate its molecular mass, we need the atomic masses of calcium, nitrogen, and oxygen from the periodic table:
- Calcium (Ca): Approximately 40.08 amu
- Nitrogen (N): Approximately 14.01 amu
- Oxygen (O): Approximately 16.00 amu
Using these values, the molecular mass of Ca(NO3)2 can be calculated as follows:
Molecular mass of Ca(NO3)2 = (1 x Atomic mass of Ca) + (2 x Atomic mass of N) + (6 x Atomic mass of O)
Molecular mass of Ca(NO3)2 = (1 x 40.08) + (2 x 14.01) + (6 x 16 Easy to understand, harder to ignore..
Molecular mass of Ca(NO3)2 = 40.08 + 28.02 + 96 And that's really what it comes down to..
Molecular mass of Ca(NO3)2 = 164.10 amu
That's why, the molecular mass of calcium nitrate [Ca(NO3)2] is approximately 164.10 amu.
Significance in Chemical Calculations
The accurate calculation of the molecular mass of calcium nitrate [Ca(NO3)2] is essential for various chemical calculations. Here's one way to look at it: if you need to prepare a specific concentration of calcium nitrate solution, knowing the molecular mass allows you to calculate the exact amount of the compound needed to dissolve in a given volume of solvent But it adds up..
In stoichiometric calculations, the molecular mass is used to convert between mass and moles, enabling you to predict the amounts of reactants and products involved in a chemical reaction. Consider this: this is particularly important in industrial processes where precise control over reaction conditions is necessary to maximize yield and minimize waste. Understanding the molecular mass of Ca(NO3)2 also helps in quantitative analysis, where the concentration of calcium nitrate in a sample is determined through techniques such as titration or spectrophotometry.
It sounds simple, but the gap is usually here Easy to understand, harder to ignore..
Trends and Latest Developments
Current Trends in Chemical Education
Modern chemical education increasingly emphasizes hands-on learning and the use of computational tools to enhance understanding of fundamental concepts like molecular mass. Interactive simulations and virtual labs allow students to visualize molecules and explore their properties in a dynamic and engaging way Worth keeping that in mind. And it works..
The integration of real-world applications into the curriculum helps students appreciate the relevance of chemistry in everyday life. As an example, case studies involving the use of calcium nitrate in agriculture or water treatment can illustrate the practical importance of understanding its molecular mass and chemical properties That's the part that actually makes a difference. Worth knowing..
Data-Driven Insights and Research
Advancements in data analytics and machine learning are transforming the way chemists analyze and interpret experimental data. These technologies can be used to predict the properties of chemical compounds, including their molecular mass, based on their structural features.
Researchers are also developing new methods for determining molecular mass with greater precision and accuracy. Also, for example, advanced mass spectrometry techniques can provide detailed information about the isotopic composition of molecules, allowing for more accurate calculations of molecular mass. These developments have significant implications for fields such as drug discovery, materials science, and environmental monitoring.
Popular Opinions and Misconceptions
One common misconception is that the molecular mass of a compound is a fixed value. In reality, the molecular mass can vary slightly depending on the isotopic composition of the elements. While the values listed on the periodic table represent the average atomic masses, the actual mass of a molecule can differ slightly due to variations in the abundance of isotopes.
Another misconception is that molecular mass is the same as formula mass. But since ionic compounds do not exist as discrete molecules, the term formula mass is more appropriate in these cases. While the terms are often used interchangeably, formula mass refers specifically to the mass of the formula unit in an ionic compound. Understanding these nuances is important for accurate and precise communication in chemistry.
The official docs gloss over this. That's a mistake.
Tips and Expert Advice
Practical Tips for Accurate Calculations
When calculating the molecular mass of calcium nitrate [Ca(NO3)2], it is essential to use accurate atomic mass values from a reliable source, such as the periodic table provided by IUPAC (International Union of Pure and Applied Chemistry). Rounding off atomic masses too early in the calculation can introduce errors, so it is best to use as many significant figures as possible.
Double-checking your calculations and paying attention to units can also help prevent mistakes. It is also good practice to use a calculator or spreadsheet to perform the calculations, especially when dealing with complex compounds or multiple steps Which is the point..
Real-World Examples and Applications
In agriculture, farmers use calcium nitrate as a fertilizer to provide plants with essential nutrients. Understanding the molecular mass of Ca(NO3)2 allows them to calculate the correct amount of fertilizer to apply to their crops, ensuring optimal growth and yield. Over- or under-application can lead to nutrient imbalances, reduced yields, and environmental damage It's one of those things that adds up. Turns out it matters..
In wastewater treatment, calcium nitrate can be used to control odors and prevent the formation of hydrogen sulfide. By understanding the stoichiometry of the reactions involved, engineers can determine the appropriate amount of calcium nitrate to add to the wastewater, effectively mitigating odor problems and improving water quality Most people skip this — try not to..
Common Mistakes to Avoid
One common mistake is forgetting to multiply the atomic mass of an element by the number of atoms present in the molecule. In the case of calcium nitrate, it is crucial to remember that there are two nitrogen atoms and six oxygen atoms in each formula unit Easy to understand, harder to ignore. But it adds up..
Another mistake is using incorrect atomic mass values. Always refer to a reliable periodic table to see to it that you are using the most accurate values. Also, be mindful of the units used in your calculations. Molecular mass is typically expressed in atomic mass units (amu), while molar mass is expressed in grams per mole (g/mol) Worth keeping that in mind..
Utilizing Online Tools and Resources
Several online tools and resources can help you calculate molecular mass quickly and accurately. Websites like WebQC and ChemAxon offer molecular weight calculators that allow you to enter the chemical formula of a compound and automatically calculate its molecular mass Worth knowing..
These tools can be particularly useful when dealing with complex molecules or when you need to perform multiple calculations. That said, it is still important to understand the underlying principles of molecular mass calculations so that you can verify the results and identify any potential errors.
FAQ
Q: What is the difference between molecular mass and molar mass?
A: Molecular mass is the mass of a single molecule, expressed in atomic mass units (amu), while molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol). Numerically, they are the same, but they differ in units and scale Most people skip this — try not to..
Basically the bit that actually matters in practice.
Q: How does isotopic abundance affect molecular mass?
A: Isotopes are atoms of the same element with different numbers of neutrons. So since isotopes have different masses, the isotopic abundance can affect the average atomic mass of an element. The values listed on the periodic table are weighted averages based on the natural abundance of each isotope Worth keeping that in mind..
Q: Can the molecular mass of a compound be determined experimentally?
A: Yes, techniques like mass spectrometry can be used to determine the molecular mass of a compound experimentally. Mass spectrometry involves ionizing molecules and separating them based on their mass-to-charge ratio, providing accurate information about their molecular mass and structure That's the part that actually makes a difference..
Q: Why is knowing the molecular mass important in chemistry?
A: Knowing the molecular mass is essential for performing accurate chemical calculations, such as determining the amount of a substance needed for a reaction or preparing solutions of a specific concentration. It is also crucial for understanding stoichiometry, quantitative analysis, and various industrial processes And that's really what it comes down to..
Short version: it depends. Long version — keep reading Simple, but easy to overlook..
Q: Where can I find reliable atomic mass values for calculating molecular mass?
A: You can find reliable atomic mass values on the periodic table provided by IUPAC (International Union of Pure and Applied Chemistry). IUPAC is the recognized authority on chemical nomenclature, terminology, and standardized atomic weights Nothing fancy..
Conclusion
Understanding the molecular mass of calcium nitrate [Ca(NO3)2] is more than just an academic exercise; it's a fundamental skill with practical applications in diverse fields. From ensuring precise fertilizer application in agriculture to enabling accurate chemical reactions in the lab, this knowledge underpins countless processes and innovations. By mastering the principles of molecular mass calculation, you empower yourself to make informed decisions and contribute meaningfully to scientific advancements.
Ready to put your knowledge into action? Here's the thing — the possibilities are endless, and the journey to mastering chemistry starts with understanding the basics. In real terms, explore online tools for molecular mass calculation, walk through real-world case studies, or even try designing your own experiment using calcium nitrate. Share your insights, ask questions, and let's continue this exploration together!