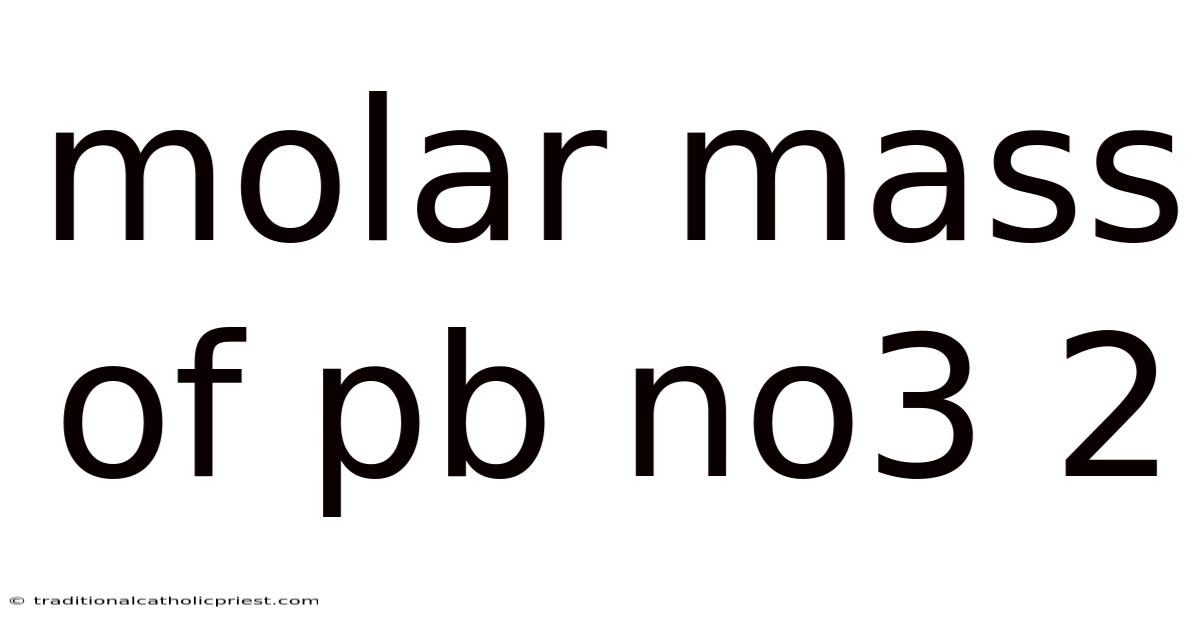Imagine you're a chemist in a bustling lab, carefully weighing out substances for a critical experiment. You need precision, accuracy, and a deep understanding of the materials you're working with. That said, one of the most fundamental concepts you rely on is molar mass – the bridge between the microscopic world of atoms and molecules and the macroscopic world of grams and kilograms that you can measure in the lab. Consider this: or perhaps you're a student grappling with stoichiometry, trying to balance equations and predict yields. The molar mass is your key to converting between grams, moles, and number of particles.
Think about the compound lead(II) nitrate, or Pb(NO3)2. It is a crystalline solid used in various applications, from pyrotechnics to mordants in dyeing. But what if you need to know exactly how much of it to use in a reaction? That's where the molar mass of Pb(NO3)2 becomes essential. This number allows you to convert between the mass you weigh out on a balance and the number of lead(II) nitrate molecules present. This article will break down calculating the molar mass of Pb(NO3)2. We'll explore its definition, understand its components, and provide a step-by-step guide to calculation. Whether you're a seasoned chemist or a curious student, you'll gain a solid understanding of this crucial concept No workaround needed..
Easier said than done, but still worth knowing Worth keeping that in mind..
Molar Mass of Pb(NO3)2: A thorough look
The molar mass of a compound is a fundamental concept in chemistry, representing the mass of one mole of that substance. A mole, in turn, is defined as 6.Consider this: 02214076 × 10^23 entities (atoms, molecules, ions, etc. ), a number known as Avogadro's constant. Thus, the molar mass connects the microscopic world of individual atoms and molecules to the macroscopic world of grams, which we can measure in the laboratory. In simpler terms, it's the mass of a specific number of particles, making it a bridge between counting individual molecules and weighing bulk quantities.
Comprehensive Overview
Understanding molar mass requires a grasp of several underlying concepts, including atomic mass, the mole concept, and chemical formulas. Let's break each of these down to build a solid foundation:
Atomic Mass: Each element on the periodic table has a unique atomic mass, which is the average mass of its atoms, taking into account the relative abundance of its isotopes. These values are typically expressed in atomic mass units (amu), but for practical purposes, we consider them to be numerically equivalent to grams per mole (g/mol) when calculating molar mass. Take this case: the atomic mass of lead (Pb) is approximately 207.2 amu, meaning that one mole of lead atoms weighs about 207.2 grams.
The Mole Concept: The mole is the SI unit for the amount of substance. It's defined as the amount of substance containing as many elementary entities as there are atoms in 12 grams of carbon-12. This number, Avogadro's constant (approximately 6.022 x 10^23), allows chemists to work with manageable quantities of atoms and molecules in the lab. One mole of any substance contains Avogadro's number of particles Turns out it matters..
Chemical Formulas: A chemical formula provides information about the types and numbers of atoms in a molecule or compound. Here's one way to look at it: the chemical formula for lead(II) nitrate is Pb(NO3)2. This tells us that each molecule of lead(II) nitrate contains one lead (Pb) atom, two nitrogen (N) atoms, and six oxygen (O) atoms. The subscript numbers indicate the number of atoms of each element present in the compound It's one of those things that adds up..
The molar mass of a compound is calculated by summing the atomic masses of all the atoms in its chemical formula. This process essentially combines the information from the periodic table (atomic masses) and the chemical formula to determine the mass of one mole of the compound No workaround needed..
Let's illustrate this with a simple example: water (H2O). Day to day, the chemical formula tells us that each water molecule contains two hydrogen atoms and one oxygen atom. The atomic mass of hydrogen is approximately 1.008 g/mol, and the atomic mass of oxygen is approximately 16.00 g/mol.
(2 × 1.008 g/mol) + (1 × 16.00 g/mol) = 18 That's the part that actually makes a difference..
What this tells us is one mole of water weighs approximately 18.016 grams. This conversion is crucial in various chemical calculations, such as determining the amount of reactants needed for a specific reaction or calculating the yield of a product Most people skip this — try not to. Less friction, more output..
Now, let's dive into the specifics of calculating the molar mass of Pb(NO3)2.
Step-by-Step Calculation of Molar Mass of Pb(NO3)2
Calculating the molar mass of Pb(NO3)2 involves breaking down the compound into its constituent elements, finding their atomic masses from the periodic table, and then summing them according to the chemical formula. Here's a detailed step-by-step guide:
Step 1: Identify the Elements and Their Quantities
The chemical formula Pb(NO3)2 indicates that one molecule of lead(II) nitrate contains:
- 1 lead (Pb) atom
- 2 nitrogen (N) atoms
- 6 oxygen (O) atoms
It's essential to correctly interpret the subscripts and parentheses in the formula. The subscript '2' outside the parentheses applies to everything inside the parentheses, meaning there are two nitrate (NO3) groups Still holds up..
Step 2: Find the Atomic Masses of Each Element
Refer to the periodic table to find the atomic masses of each element. Use a reliable periodic table source, such as a textbook, reputable website, or chemistry software. The atomic masses are:
- Lead (Pb): 207.2 g/mol
- Nitrogen (N): 14.01 g/mol
- Oxygen (O): 16.00 g/mol
Note that these values are rounded to two decimal places for simplicity. Depending on the required precision, you may need to use more decimal places Easy to understand, harder to ignore. That's the whole idea..
Step 3: Multiply the Atomic Mass by the Number of Atoms
Multiply the atomic mass of each element by the number of atoms of that element in the compound:
- Lead (Pb): 1 atom × 207.2 g/mol = 207.2 g/mol
- Nitrogen (N): 2 atoms × 14.01 g/mol = 28.02 g/mol
- Oxygen (O): 6 atoms × 16.00 g/mol = 96.00 g/mol
This step calculates the total mass contribution of each element to the molar mass of the compound.
Step 4: Sum the Results
Add up the results from the previous step to get the molar mass of Pb(NO3)2:
Molar mass of Pb(NO3)2 = 207.Now, 2 g/mol (Pb) + 28. 02 g/mol (N) + 96.00 g/mol (O) = 331 And that's really what it comes down to. And it works..
So, the molar mass of lead(II) nitrate (Pb(NO3)2) is approximately 331.Which means 22 g/mol. What this tells us is one mole of Pb(NO3)2 weighs about 331.22 grams Still holds up..
Trends and Latest Developments
While the fundamental principles of calculating molar mass remain constant, there are some trends and developments to consider:
Increased Precision in Atomic Mass Values: Atomic mass values are constantly being refined as measurement techniques improve. Organizations like the International Union of Pure and Applied Chemistry (IUPAC) regularly update the official atomic mass values of elements. What this tells us is the molar mass of compounds may also change slightly over time as more accurate atomic mass data become available It's one of those things that adds up..
Use of Software and Online Calculators: Numerous software tools and online molar mass calculators are available to simplify the calculation process. These tools can automatically look up atomic masses, perform the calculations, and even account for isotopic abundances. While these tools are convenient, it's still crucial to understand the underlying principles of molar mass calculation It's one of those things that adds up. Simple as that..
Importance in Nanotechnology: Molar mass calculations are becoming increasingly important in nanotechnology, where precise control over the stoichiometry of materials is essential. Nanomaterials often have unique properties that depend on their size, shape, and composition. Accurate molar mass calculations are needed to synthesize these materials with the desired properties Simple as that..
Applications in Environmental Chemistry: Molar mass calculations are also vital in environmental chemistry for determining the concentrations of pollutants and contaminants in various samples. To give you an idea, knowing the molar mass of a specific pollutant allows scientists to convert between mass concentrations (e.g., mg/L) and molar concentrations (e.g., mol/L), which are essential for assessing environmental risks and designing remediation strategies.
Tips and Expert Advice
Here are some practical tips and expert advice to help you master molar mass calculations and avoid common mistakes:
Double-Check the Chemical Formula: confirm that you have the correct chemical formula for the compound you are working with. A mistake in the formula will lead to an incorrect molar mass calculation. Pay close attention to subscripts and parentheses, and verify the formula with a reliable source Easy to understand, harder to ignore..
Use Accurate Atomic Mass Values: Use atomic mass values from a reliable periodic table source. Avoid using outdated or inaccurate values, as this can affect the accuracy of your results. Always check the source of your atomic mass values and use the most up-to-date information available Worth keeping that in mind..
Pay Attention to Units: Always include the correct units (g/mol) when reporting molar mass values. This helps to avoid confusion and ensures that your results are interpreted correctly. Consistent use of units is crucial for dimensional analysis and other chemical calculations Less friction, more output..
Practice Regularly: The best way to master molar mass calculations is to practice regularly. Work through various examples and problems, and try to solve them without using a calculator or online tool. This will help you develop a deeper understanding of the concepts and improve your problem-solving skills.
Understand the Significance of Molar Mass: Don't just memorize the steps for calculating molar mass; understand why it is important. Molar mass is a fundamental concept in chemistry that is used in many different types of calculations. Understanding its significance will help you appreciate its importance and apply it correctly in various contexts.
Use Molar Mass in Stoichiometry: Stoichiometry is the calculation of quantitative relationships in chemical reactions. Molar mass is essential for converting between grams and moles in stoichiometric calculations. As an example, you can use molar mass to determine the amount of reactants needed to produce a specific amount of product or to calculate the percent yield of a reaction.
By following these tips and expert advice, you can improve your understanding of molar mass calculations and apply them confidently in various chemical applications Most people skip this — try not to..
FAQ
Q: What is the difference between atomic mass and molar mass?
A: Atomic mass refers to the mass of a single atom of an element, typically expressed in atomic mass units (amu). In practice, molar mass, on the other hand, is the mass of one mole (6. 022 x 10^23) of atoms, molecules, or ions of a substance, expressed in grams per mole (g/mol). The numerical value of atomic mass is the same as the molar mass, but they have different units and refer to different quantities.
Q: Can the molar mass of a compound be a decimal?
A: Yes, the molar mass of a compound can be a decimal. This is because atomic masses are average values that take into account the relative abundance of different isotopes of an element. When you sum the atomic masses of all the atoms in a compound, the result is often a decimal value Small thing, real impact..
Q: How does molar mass relate to molecular weight?
A: The terms "molar mass" and "molecular weight" are often used interchangeably, but there is a subtle difference. Plus, molecular weight is the mass of a single molecule expressed in atomic mass units (amu), while molar mass is the mass of one mole of molecules expressed in grams per mole (g/mol). Numerically, they are the same, but they have different units and refer to different quantities And that's really what it comes down to. That's the whole idea..
Real talk — this step gets skipped all the time.
Q: Why is molar mass important in chemistry?
A: Molar mass is essential in chemistry because it provides a way to convert between mass and moles. Which means this conversion is crucial for various chemical calculations, such as determining the amount of reactants needed for a specific reaction, calculating the yield of a product, and determining the concentration of a solution. Molar mass allows chemists to work with manageable quantities of atoms and molecules in the lab and to make accurate predictions about chemical reactions Still holds up..
Q: Where can I find reliable atomic mass values?
A: You can find reliable atomic mass values in several sources, including:
- The periodic table: Most periodic tables include atomic mass values for each element.
- Chemistry textbooks: Chemistry textbooks typically have a table of atomic masses in the appendix.
- Reputable websites: Websites like the National Institute of Standards and Technology (NIST) and the International Union of Pure and Applied Chemistry (IUPAC) provide accurate atomic mass data.
- Chemistry software: Many chemistry software programs include a built-in database of atomic masses.
Conclusion
In a nutshell, the molar mass of Pb(NO3)2 is approximately 331.Now, 22 g/mol. This value is crucial for various chemical calculations involving lead(II) nitrate, from preparing solutions to performing stoichiometric analyses. By understanding the concepts of atomic mass, the mole, and chemical formulas, you can confidently calculate the molar mass of any compound Still holds up..
This changes depending on context. Keep that in mind Simple, but easy to overlook..
Now that you've gained a solid understanding of molar mass and its calculation, put your knowledge to the test! Dive deeper into stoichiometry, solution chemistry, and chemical kinetics to see how this fundamental concept underpins many essential calculations. In practice, try calculating the molar masses of other compounds and explore how molar mass is used in different areas of chemistry. Plus, what other chemical compounds pique your interest? Share your newfound knowledge with peers, discuss challenging problems, and continue to explore the fascinating world of chemistry. Let us know in the comments below, and let's continue this journey of chemical discovery together.