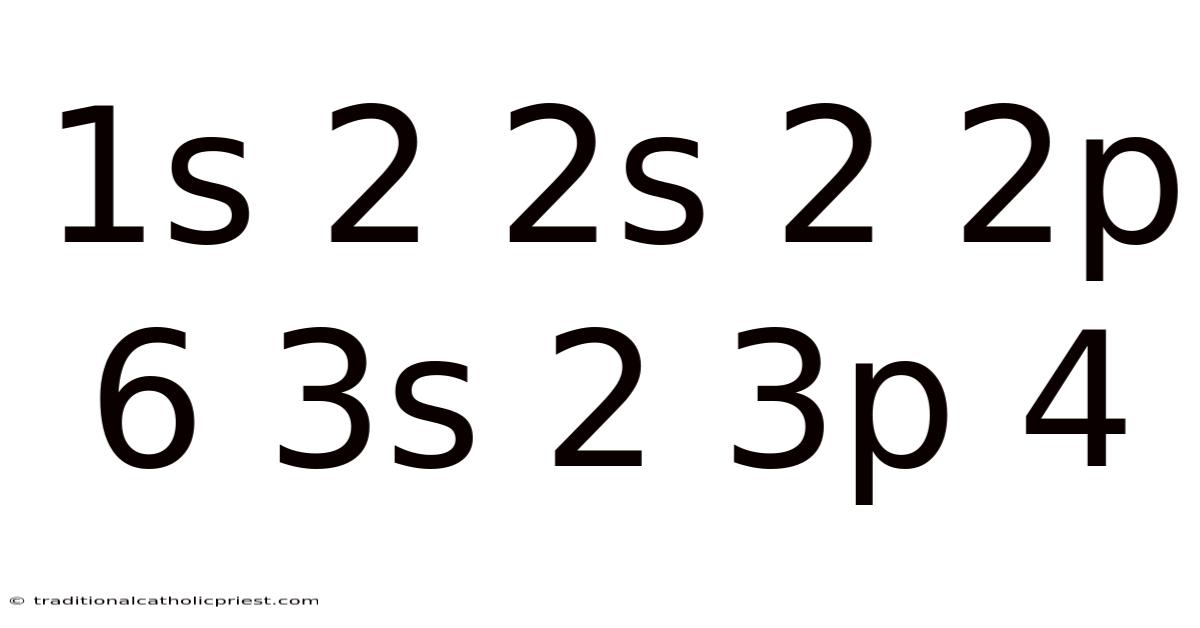Imagine walking into a bustling marketplace, each stall displaying a unique arrangement of goods. Just as a merchant carefully organizes their wares, atoms arrange their electrons in specific patterns. These arrangements aren't random; they follow precise rules dictated by the principles of quantum mechanics. One of the most fundamental ways to describe these arrangements is through electron configurations, like the seemingly cryptic "1s² 2s² 2p⁶ 3s² 3p⁴".
This notation might look intimidating at first glance, but it's actually a concise and powerful way to represent the electronic structure of an atom. It tells us exactly how many electrons reside in each energy level and subshells around the nucleus. Understanding electron configurations unlocks a deeper understanding of an element's chemical properties, its reactivity, and how it interacts with other atoms to form molecules. In this full breakdown, we'll break down the components of electron configurations, explore the rules governing their construction, and see how they relate to the periodic table and the behavior of elements. So, let's embark on this journey to demystify the seemingly complex world of electron configurations, starting with the key example: 1s² 2s² 2p⁶ 3s² 3p⁴.
Unveiling the Meaning of 1s² 2s² 2p⁶ 3s² 3p⁴: A Deep Dive into Electron Configuration
To truly grasp the meaning of the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴, it's essential to understand the fundamental concepts that underpin it. This notation describes how electrons are distributed within the different energy levels and sublevels (also known as subshells) within an atom. Each part of the notation reveals specific information about the location and number of electrons Easy to understand, harder to ignore..
The numbers (1, 2, 3, etc.Higher numbers indicate energy levels that are further away from the nucleus and have higher energy. The letters (s, p, d, f) denote the subshell, which describes the shape of the electron's orbital and also corresponds to a slightly different energy level within a given shell. ) represent the principal energy level, also known as the electron shell. The superscripts (², ⁶, ⁴) indicate the number of electrons occupying that particular subshell.
In our example, 1s² 2s² 2p⁶ 3s² 3p⁴, the "1" indicates the first energy level (closest to the nucleus), and the "s" indicates the s subshell. So the superscript "2" tells us that there are two electrons in the 1s subshell. This leads to similarly, the "2s²" indicates two electrons in the s subshell of the second energy level. And the "2p⁶" means six electrons in the p subshell of the second energy level. Finally, "3s² 3p⁴" tells us that the third energy level has two electrons in the s subshell and four electrons in the p subshell. By adding up the superscripts (2 + 2 + 6 + 2 + 4 = 16), we can determine the total number of electrons in the atom, which in this case is 16. This corresponds to the element sulfur (S).
A Comprehensive Overview of Electron Configuration
To fully comprehend the concept of electron configurations, let's delve deeper into the fundamental principles that govern them. These principles stem from quantum mechanics and provide the rules for how electrons arrange themselves around the nucleus of an atom.
1. Orbitals and Quantum Numbers: Electrons don't orbit the nucleus in neat, predictable paths like planets around a star. Instead, they exist in regions of space called orbitals, which are described by a set of four quantum numbers:
- Principal Quantum Number (n): As mentioned earlier, this number defines the energy level or electron shell. It can be any positive integer (n = 1, 2, 3, ...). Higher values of n correspond to higher energy levels and greater average distances from the nucleus.
- Angular Momentum or Azimuthal Quantum Number (l): This number describes the shape of the orbital and defines the subshell. It can range from 0 to n-1. l = 0 corresponds to an s subshell (spherical shape), l = 1 to a p subshell (dumbbell shape), l = 2 to a d subshell (more complex shape), and l = 3 to an f subshell (even more complex shape).
- Magnetic Quantum Number (ml): This number describes the orientation of the orbital in space. It can take on integer values from -l to +l, including 0. Take this: a p subshell (l = 1) has three possible orientations (ml = -1, 0, +1), corresponding to three p orbitals oriented along the x, y, and z axes.
- Spin Quantum Number (ms): This number describes the intrinsic angular momentum of an electron, which is quantized and called spin. Electrons behave as if they are spinning, creating a magnetic dipole moment. The spin quantum number can be either +1/2 (spin up) or -1/2 (spin down).
2. The Pauli Exclusion Principle: This fundamental principle states that no two electrons in the same atom can have the same set of all four quantum numbers. In simpler terms, each orbital can hold a maximum of two electrons, and these two electrons must have opposite spins. This principle is crucial for understanding the arrangement of electrons in atoms.
3. Hund's Rule: When filling degenerate orbitals (orbitals with the same energy, such as the three p orbitals in a p subshell), electrons will individually occupy each orbital before doubling up in any one orbital. To build on this, all of these singly occupied orbitals will have the same spin (maximizing the total spin). This rule minimizes electron-electron repulsion and leads to a more stable electron configuration.
4. The Aufbau Principle: This principle provides a guideline for filling orbitals in the correct order of energy. In general, electrons first fill the lowest energy orbitals available before moving to higher energy orbitals. The filling order is typically: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p. Notice that the 4s orbital is filled before the 3d orbital, and the 5s orbital is filled before the 4d orbital. This is because the energy levels of orbitals can overlap, and the 4s orbital has a slightly lower energy than the 3d orbital in many atoms. A helpful mnemonic device for remembering this order is the diagonal rule.
5. Exceptions to the Aufbau Principle: While the Aufbau principle provides a useful guideline, there are exceptions to it, particularly for transition metals. These exceptions arise due to the stability associated with having either a completely filled or half-filled d subshell. To give you an idea, chromium (Cr) has an expected electron configuration of [Ar] 4s² 3d⁴. Even so, its actual electron configuration is [Ar] 4s¹ 3d⁵. By promoting one electron from the 4s orbital to the 3d orbital, chromium achieves a half-filled 3d subshell, which is a more stable configuration. Similarly, copper (Cu) has an expected electron configuration of [Ar] 4s² 3d⁹, but its actual electron configuration is [Ar] 4s¹ 3d¹⁰, resulting in a completely filled 3d subshell.
6. Noble Gas Configuration: To simplify writing electron configurations, we can use the noble gas configuration. This involves using the symbol of the noble gas that precedes the element in brackets, followed by the remaining electron configuration. To give you an idea, the electron configuration of sulfur (S) is 1s² 2s² 2p⁶ 3s² 3p⁴. Neon (Ne) has an electron configuration of 1s² 2s² 2p⁶. Because of this, we can write the electron configuration of sulfur as [Ne] 3s² 3p⁴.
Trends and Latest Developments in Electron Configuration Research
Electron configurations are not just a static concept taught in introductory chemistry courses. They are a dynamic area of research that continues to evolve with advancements in computational chemistry and experimental techniques.
1. Computational Chemistry: Sophisticated computational methods, such as density functional theory (DFT) and ab initio calculations, are used to predict and refine electron configurations, especially for complex molecules and materials. These methods can account for electron correlation effects and relativistic effects, which are important for heavier elements.
2. Experimental Techniques: Experimental techniques, such as X-ray photoelectron spectroscopy (XPS) and Auger electron spectroscopy (AES), are used to experimentally determine the electron configurations of materials. These techniques provide valuable information about the elemental composition and chemical bonding of surfaces and interfaces.
3. High-Throughput Screening: With the advent of high-throughput computing, researchers are using electron configurations as a starting point for screening new materials with desired properties. By computationally generating and analyzing the electron configurations of thousands of compounds, they can identify promising candidates for applications such as solar cells, catalysts, and battery materials That's the part that actually makes a difference..
4. Understanding Chemical Reactivity: Electron configurations play a central role in understanding chemical reactivity. By analyzing the electron configurations of reactants, chemists can predict the likelihood and mechanism of a chemical reaction. To give you an idea, the electron configuration of an atom determines its electronegativity, which is a measure of its ability to attract electrons in a chemical bond Took long enough..
5. Quantum Computing: The emergence of quantum computing offers the potential to revolutionize the calculation of electron configurations. Quantum computers can efficiently solve complex quantum mechanical problems that are intractable for classical computers, opening up new avenues for understanding the electronic structure of matter.
Tips and Expert Advice for Mastering Electron Configurations
Understanding electron configurations is crucial for success in chemistry and related fields. Here are some tips and expert advice to help you master this important concept:
1. Practice, Practice, Practice: The best way to learn electron configurations is to practice writing them for a variety of elements and ions. Start with simple elements and gradually work your way up to more complex ones. Use the periodic table as a guide and refer to the Aufbau principle and Hund's rule Not complicated — just consistent..
2. Use the Periodic Table as a Cheat Sheet: The periodic table is organized in a way that reflects the electron configurations of the elements. The group number (vertical column) indicates the number of valence electrons (electrons in the outermost shell), and the period number (horizontal row) indicates the principal energy level of the valence electrons.
3. Learn the Exceptions: Be aware of the exceptions to the Aufbau principle, particularly for transition metals. Remember that atoms strive for stability, and achieving a completely filled or half-filled d subshell can lead to a more stable electron configuration Took long enough..
4. Draw Orbital Diagrams: Orbital diagrams are a visual way to represent electron configurations. They show the individual orbitals within each subshell and the spin of each electron. Drawing orbital diagrams can help you understand Hund's rule and the Pauli exclusion principle.
5. Understand the Relationship to Chemical Properties: Connect electron configurations to the chemical properties of elements. Take this: elements with similar valence electron configurations tend to have similar chemical behavior. The number of valence electrons determines an element's ability to form chemical bonds.
6. Use Online Resources: There are many online resources available to help you learn electron configurations, including tutorials, practice quizzes, and interactive simulations. Take advantage of these resources to reinforce your understanding.
7. Focus on the Underlying Principles: Don't just memorize the rules for writing electron configurations. Focus on understanding the underlying principles of quantum mechanics that govern the arrangement of electrons in atoms. This will give you a deeper and more lasting understanding of the concept.
8. Work Through Examples with a Tutor or Classmate: Sometimes, the best way to understand a difficult concept is to work through examples with someone else. Explain the concepts to each other and help each other understand the reasoning behind each step No workaround needed..
9. Relate Electron Configurations to Real-World Applications: Understanding electron configurations is not just an academic exercise. It has many real-world applications, such as designing new materials, developing new technologies, and understanding chemical reactions. By relating electron configurations to real-world applications, you can see the relevance and importance of this concept Worth knowing..
10. Don't Be Afraid to Ask Questions: If you are struggling to understand electron configurations, don't be afraid to ask questions. Ask your teacher, your tutor, or your classmates for help. There are no stupid questions, and asking questions is a sign that you are engaged in the learning process It's one of those things that adds up..
Frequently Asked Questions (FAQ) About Electron Configurations
Q: What is the difference between an electron configuration and an orbital diagram?
A: An electron configuration is a shorthand notation that shows the distribution of electrons among the various energy levels and subshells in an atom. An orbital diagram is a visual representation that shows the individual orbitals within each subshell and the spin of each electron Easy to understand, harder to ignore. Worth knowing..
Q: Why are electron configurations important?
A: Electron configurations are important because they determine the chemical properties of elements. They explain how atoms interact with each other to form chemical bonds and predict the reactivity of different substances The details matter here. That alone is useful..
Q: What are valence electrons?
A: Valence electrons are the electrons in the outermost shell of an atom. They are the electrons that are involved in chemical bonding and determine the chemical properties of an element.
Q: How do you determine the number of valence electrons from an electron configuration?
A: To determine the number of valence electrons, identify the highest principal energy level (n) in the electron configuration. The valence electrons are the electrons in that energy level. Here's one way to look at it: in the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴, the highest principal energy level is 3. There are two electrons in the 3s subshell and four electrons in the 3p subshell, so there are a total of six valence electrons.
Q: What are the exceptions to the Aufbau principle?
A: The exceptions to the Aufbau principle occur when atoms can achieve a more stable electron configuration by promoting an electron from a lower energy level to a higher energy level. This typically happens when atoms can achieve a completely filled or half-filled d subshell Small thing, real impact. That's the whole idea..
Q: How do you write the electron configuration of an ion?
A: To write the electron configuration of an ion, first write the electron configuration of the neutral atom. Then, add or remove electrons to account for the charge of the ion. For a positive ion (cation), remove electrons from the highest energy level first. For a negative ion (anion), add electrons to the lowest energy level available It's one of those things that adds up..
Q: Can electron configurations be used to predict the properties of molecules?
A: Yes, electron configurations can be used to predict the properties of molecules. By understanding the electron configurations of the atoms in a molecule, chemists can predict the types of bonds that will form, the shape of the molecule, and its reactivity And it works..
Conclusion
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴, representing sulfur, is more than just a string of symbols; it's a roadmap to understanding an atom's behavior. Also, by mastering the principles of electron configurations, including the Aufbau principle, Hund's rule, and the Pauli exclusion principle, you gain a powerful tool for predicting chemical properties and understanding the world around you. Remember to practice writing electron configurations, use the periodic table as a guide, and focus on the underlying principles Most people skip this — try not to. Which is the point..
Ready to put your knowledge to the test? Explore online resources and interactive simulations to reinforce your understanding. And don't hesitate to ask questions and seek help when needed. Try writing the electron configurations for other elements and ions. By mastering electron configurations, you'll access a deeper understanding of the fascinating world of chemistry The details matter here..